Description
Photocatalyzed reactions are a particular class of photochemical processes that occur only in the presence of a catalyst and upon irradiation with light. Photocatalysis was first introduced as a method for the production of hydrogen from water (water splitting) or for the photodepollution of water or air. Nevertheless, in recent years this approach has also been used for the synthesis of organic molecules. [1-3]
Commonly used photocatalysts are:
- Solid semiconductor particles (e.g. TiO2);
- Organic molecules (e.g. benzophenone, cianoaromatics, dyes);
- Inorganic salts (e.g. decatungstate anion).
The main features of photocatalysis are:
- very mild reaction conditions. The use of the (highly reactive) excited state of the photocatalyst avoids the use of any aggressive auxiliary material or the use of high temperatures; this notwithstanding, the method generates radicals from unusual, non activated precursors, such as alkanes and amides;
- Contrary to the usual photochemical approach, which requires the use of light absorbing reagents, the photocatalytic method can be used to activate molecules virtually photochemically unreactive (e.g. alkanes), since the photocatalyst is the only absorbing light species in the reaction mixture.
It is important to stress that all the features previously reported well fit with the main principles of green chemistry. Photocatalysis is thus a really environment-friendly candidate for a valid alternative to the known thermal synthetic strategies.
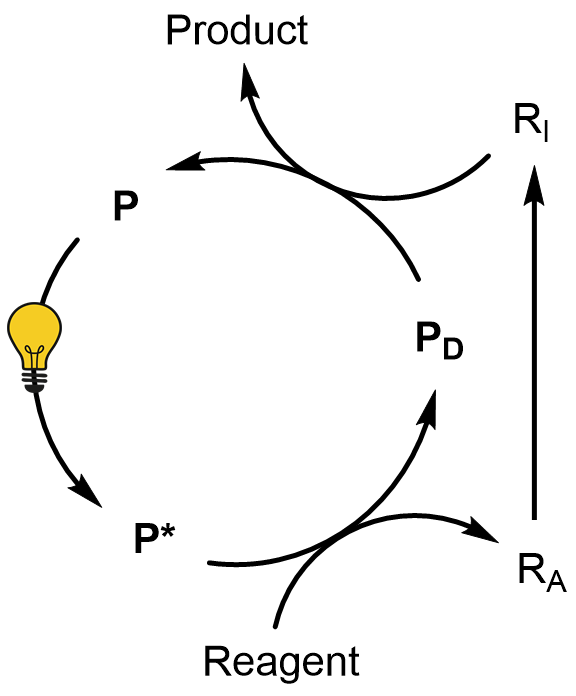 The term photocatalysis comes from the union of the words “photochemistry” and “catalysis”. On one hand, the photocatalyst P absorbs the radiation and is chemically active in the excited state, on the other hand it is a part of a catalytic cycle and can be used in a sub-stoichiometric amount, as it happens in thermal catalysis. As for the above, the photocatalytic method (see the Scheme on the left) is based on the use of the photoactive compound P.
The term photocatalysis comes from the union of the words “photochemistry” and “catalysis”. On one hand, the photocatalyst P absorbs the radiation and is chemically active in the excited state, on the other hand it is a part of a catalytic cycle and can be used in a sub-stoichiometric amount, as it happens in thermal catalysis. As for the above, the photocatalytic method (see the Scheme on the left) is based on the use of the photoactive compound P.
This is able to absorb light and, once reached the excited state (P*), it is able to activate (usually by hydrogen abstraction or electron transfer) the reagent (R). The resulting intermediate (RA) then evolves to RI and, after reaction with the deactivated photocatalyst (PD), gives the end product with the concomitant regeneration of P.
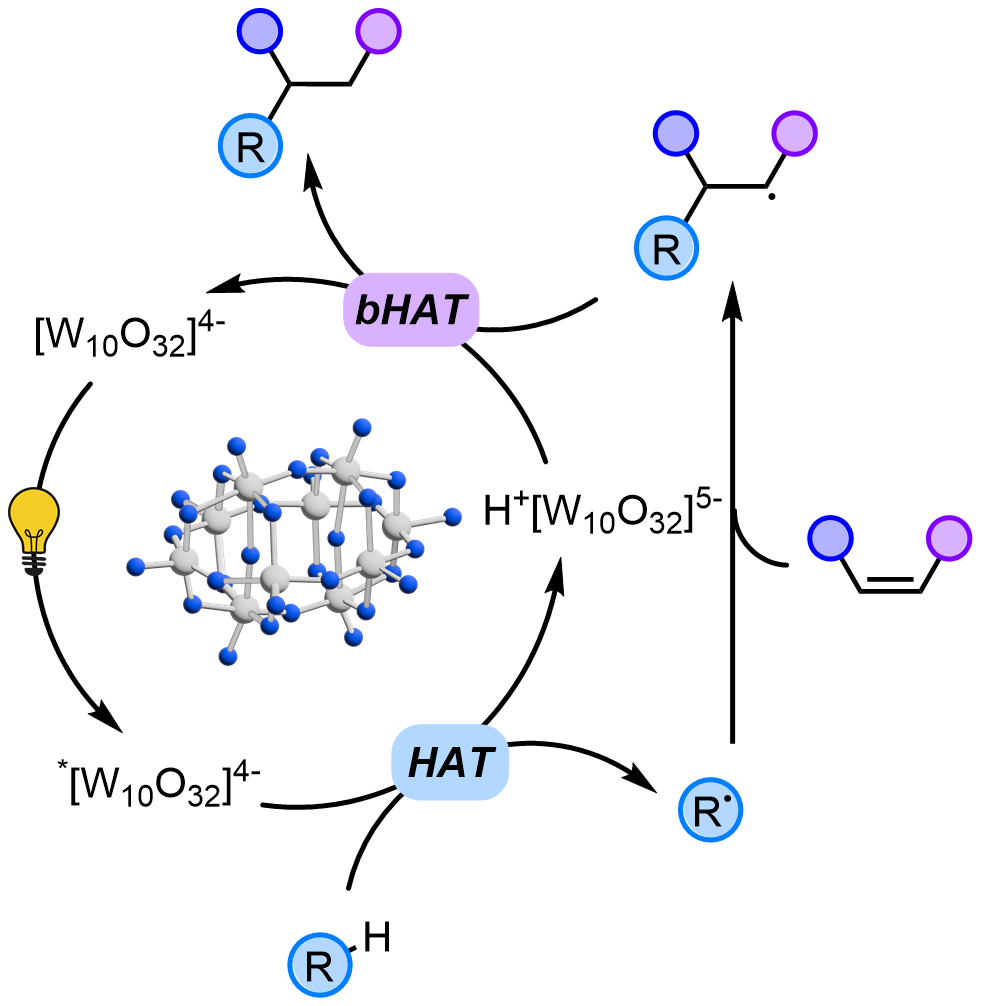
Recently, our research group has focused its attention on TBADT (tetrabutylammonium decatungstate). This compound (a white powder) is able to activate the C-H bonds in various molecules by homolytic cleavage of this bond (see the Scheme below) and allows for the generation of radicals (R•) under very mild conditions. The latter intermediate is then trapped by electron-poor olefins (α,β-unsaturated esters, ketones or nitriles) resulting in an overall radical conjugate addition onto a C=C double bond.
TBADT has proven to be a robust and versatile photocatalyst: it can be used in a small amount (2% eqv.) and remains active for many catalytic cycles. Moreover, it is able to activate many different classes of substrates, otherwise non-reactive. M
any compounds have been used as substrates for this reaction: alkanes, alcohols, ethers, acetals, aldehydes and amides. [4]
Future developments
Recently, our attention has been focused on the axploitation of the decatungstate photocatalyst for photoinduced electron transfer reactions (PET, in alternative to the above mentioned hydrogen atom transfer – HAT- mechanism). [5] Another field under investigation concerns with the discovery of innovative photocatalysts able to work under visible light irradiation.
References
- Fagnoni, M.; Dondi, D.; Ravelli, D.; Albini, A. “Photocatalysis for the Formation of the C-C Bond” Chem. Rev. 2007, 107, 2725-2756. DOI: 10.1021/cr068352x
- Ravelli, D.; Dondi, D.; Fagnoni, M.; Albini, A. “Photocatalysis. A multi-faceted concept for green chemistry” Chem. Soc. Rev. 2009, 38, 1999-2011. DOI: 10.1039/B714786B
- Ravelli, D.; Fagnoni, M.; Albini, A. “Photoorganocatalysis. What for?” Chem. Soc. Rev. 2013, 42, 97-113. DOI: 10.1039/C2CS35250H
- Ravelli, D.; Montanaro, S.; Zema, M.; Fagnoni, M.; Albini, A. “A Tin-Free, Radical Photocatalyzed Addition to Vinyl Sulfones” Adv. Synth. Catal. 2011, 353, 3295-3300 and references therein. DOI: 10.1002/adsc.201100591
- Montanaro, S.; Ravelli, D.; Merli, D.; Fagnoni, M.; Albini, A. “Decatungstate As Photoredox Catalyst: Benzylation of Electron-Poor Olefins” Org. Lett. 2012, 14, 4218-4221. DOI: 10.1021/ol301900p
